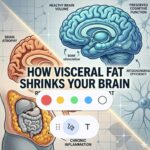
Relationship Between Visceral Fat and Brain Health
Recent longitudinal research from the FIT project indicates that visceral adipose tissue (VAT)—the metabolically active fat surrounding internal organs—is a primary modifiable risk factor for neurodegeneration.
-
Brain Atrophy: Reductions in visceral fat are directly linked to higher total brain volume, increased gray matter, and improved hippocampal occupancy scores.
-
Cognitive Performance: Lower cumulative exposure to visceral fat over a decade correlates with higher scores on the Montreal Cognitive Assessment (MoCA), particularly in memory indices.
-
VAT vs. BMI: Unlike visceral fat, changes in BMI and subcutaneous fat showed no significant association with brain structure or atrophy rates.
-
The “Canary” Effect: Cognitive decline often precedes visible brain shrinking in individuals with high visceral fat, serving as an early warning sign of neurodegeneration.
High Visceral Fat = Smaller Brain
The phenomenon of brain shrinkage—medically termed cerebral atrophy—is a natural part of aging, but the rate at which it occurs is highly variable and heavily influenced by metabolic and lifestyle factors. While the average adult loses approximately 0.2% to 0.5% of brain volume per year starting in their 30s, this rate can accelerate significantly under specific conditions, especially high VAT.
Key Regions Vulnerable to Shrinkage
-
Hippocampus: Crucial for memory formation. This area is often the first to show significant atrophy in response to chronic stress, high cortisol, and poor glycemic control.
-
Prefrontal Cortex: Responsible for executive function, decision-making, and focus. Shrinkage here often manifests as “brain fog” or decreased cognitive flexibility.
-
Gray Matter vs. White Matter: Gray matter contains the neuronal cell bodies, while white matter consists of the “cables” (axons) connecting them. Atrophy in gray matter directly impacts processing power, while white matter decay slows down communication speed between brain regions.
Beyond Visceral Fat: Other Drivers of Atrophy
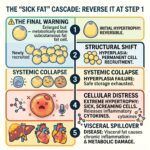
While the FIT project highlighted visceral fat, other technical factors play a major role in cellular-level brain shrinkage:
-
The Cell Danger Response (CDR): When mitochondria sense environmental or metabolic threats, they shift from energy production to a defensive mode. If mitochondria stay in this “shielding” state too long, it leads to cellular senescence and eventual tissue loss in the brain.
-
Glycemic Variability: Large spikes and crashes in blood glucose (even within “normal” ranges) cause micro-vascular damage. Since the brain is the most glucose-demanding organ, these fluctuations lead to localized energy crises and neuronal death.
-
NAD+ Depletion: Nicotinamide adenine dinucleotide (NAD+) is essential for DNA repair and mitochondrial function. As levels drop with age, the brain’s ability to repair oxidative damage diminishes, accelerating volume loss.
-
Alcohol Consumption: Chronic alcohol use is a potent neurotoxin that causes immediate and long-term volume loss in the frontal lobes and cerebellum.
Mitigating Atrophy and Promoting Neurogenesis
Maintaining “Peakspan”—90% of peak function—requires strategies that counteract the biological “rusting” of brain tissue.
-
Urolithin A and Autophagy: Compounds like Urolithin A (Mitopure) support mitophagy—the clearing out of damaged mitochondria—allowing for more efficient energy production in neurons.
-
BDNF Stimulation: Brain-Derived Neurotrophic Factor is essentially “fertilizer” for the brain. Vigorous aerobic exercise and high-intensity training are the most potent ways to spike BDNF, which can actually help grow new neurons in the hippocampus.
-
NAD+ Optimization: Prioritizing NAD+ precursors (e.g., nicotinamide) or lifestyle habits that preserve NAD+ (e.g., fasting or specific exercise protocols) supports SIRT1 enzymes that protect brain cells from aging.
-
Hypoxia and Nitric Oxide: Brief, controlled bouts of low oxygen (like certain breathing techniques or specific HIIT methods), and nose breathing (increases N.O.) can improve vascular health and blood flow to deep brain structures.
Biological Mechanisms
The study investigated several pathways to determine how deep abdominal fat impacts the brain.
-
Glycemic Control: Fasting blood glucose and HbA1c were identified as the primary drivers. Neuroprotective benefits from fat loss are largely achieved through improved glucose regulation.
-
Inflammatory Markers: Systemic inflammation (CRP) and blood lipid levels were not identified as the primary mechanisms in this study.
Evidence-Based Strategies for Reduction
Visceral fat is highly responsive to lifestyle interventions due to its high metabolic activity.
-
Calorie Deficit: A sustained energy deficit is required. Consistency in the deficit is the primary driver of VAT loss, regardless of specific macronutrient ratios.
-
Aerobic Exercise: This is the most effective modality for targeting visceral fat. High-intensity interval training (HIIT) and vigorous aerobic sessions outperform resistance training for direct reduction, although X Gym style strength training produces similar VAT los results as HIIT aerobic training since it creates the same heart rate “Spikes and Valleys” effect.
-
Sleep Hygiene: Chronic sleep restriction (e.g., 4 hours per night) increases ghrelin and cortisol, specifically promoting visceral fat accumulation.
-
Consistent Activity: Weekend warriors won’t see the same significant impact as those who exercise throughout the week.
Practical Monitoring
Since specialized devices like BEI scales, MRI, or DEXA scans are not always accessible, indirect measures can track progress:
-
Belly Circumference (measured at the navel): A reliable proxy for VAT.
-
Risk Thresholds: Increased risk is noted at circumferences of <36 inches for men and <32 inches for women.